|
Back to Blog
Ionic Bonds: Ionic bonds have a low conductivity.Ĭovalent Bonds: Covalent bonds have a very low conductivity. Metallic Bonds:Metallic bonds form when a variable number of atoms share a variable number of electrons in a metal lattice. Ionic Bonds: Ionic bonds form when one atom provides electrons to another atom.Ĭovalent Bonds:Covalent bonds form when two atom shares their valence electrons. Metallic Bonds:Bond Energy is lower than other primary bonds. Ionic Bonds: Bond Energy is higher than metallic bonds.Ĭovalent Bonds:Bond Energy is higher than metallic bonds. Metallic bond:Metallic bonds are forces between negatively charged freely moving electrons and positively charged metal ions. Ionic bond: Ionic bonds are electrostatic forces arising between negative and positive ions.Ĭovalent bond:Covalent bonds are bonds that occur when two elements share a valence electron in order to get electron configuration of neutral gasses. Difference Between Ionic Covalent and Metallic Bonds Definition Examples of metals with metallic bonds include iron, copper, gold, silver, nickel etc. Due to the free moving electrons, metals are known for good electricity conductors. Because of this reason, electrons in metallic bonds are referred to as delocalized electrons. In metallic bonds, almost every atom in the metal lattice shares electrons so there is no way to determine which atom shares which electron. Electrostatic forces are formed due to the attractions between the electron cloud and ions. These positively charged ions are surrounded by a large number of negatively charged, free moving electrons called an electron cloud. Once these electrons detach, metal atoms become positively charged ions. Thus, valence electrons require very low energy to release themselves from nuclei. In a metal lattice, valence electrons are loosely attached by the nuclei of metal atoms. Examples of covalently bonded material include hydrogen gas, nitrogen gas, water molecules, diamond, silica etc. Covalently bonded materials are found in all three states i.e., solid, liquid and gas. For example, fluorine needs one electron to complete its outer shell, thus, one electron is shared by another fluorine atom by making a covalent bond resulting F 2 molecule. Covalent bonds occur between same atoms or different types of atoms. The two atoms have a small difference in electronegativity. Examples of ionically bonded materials include LiF, NaCl, BeO, CaF 2 etc.Ĭovalent bonds are formed when two atoms share their valence electrons. Bonds usually occur between metal and non-metal that are having a large difference in electronegativity. 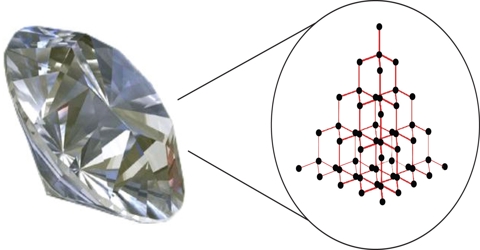
Solids bonded with ionic bonds have crystalline structures and low electrical conductivity, which is due to lack of free moving electrons. These stable bonds are also called electrostatic bonds. When these ions are brought together, the attraction forces are occurred due to opposite charges of ions. Atoms with very few electrons in their outermost shell tend to donate the electrons and become positively charged ions, while atoms with more electrons in their outermost orbit have a tendency to receive electrons and become positively charged ions. What is the difference between Ionic Covalent and Metallic Bonds? What are Ionic BondsĬertain atoms tend to donate or receive electrons in order to become more stable by completely occupying their outermost orbit. The main difference between ionic covalent and metallic bonds is their formation ionic bonds form when one atom provides electrons to another atom whereas covalent bonds form when two atom shares their valence electrons and metallic bonds form when a variable number of atoms share a variable number of electrons in a metal lattice.Ĥ. Primary bonds have relatively high bond energies and are more stable when compared with secondary forces. Secondary bonds include dispersion bonds, dipole bonds, and hydrogen bonds. There are three types of primary bonds namely ionic bonds, covalent bonds, and metallic bonds. Primary bonds are the chemical bonds that hold atoms in molecules, whereas secondary bonds are the forces that hold molecules together. Main Difference – Ionic vs Covalent vs Metallic Bondsīonds can be divided into two broad categories primary bonds and secondary bonds.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
